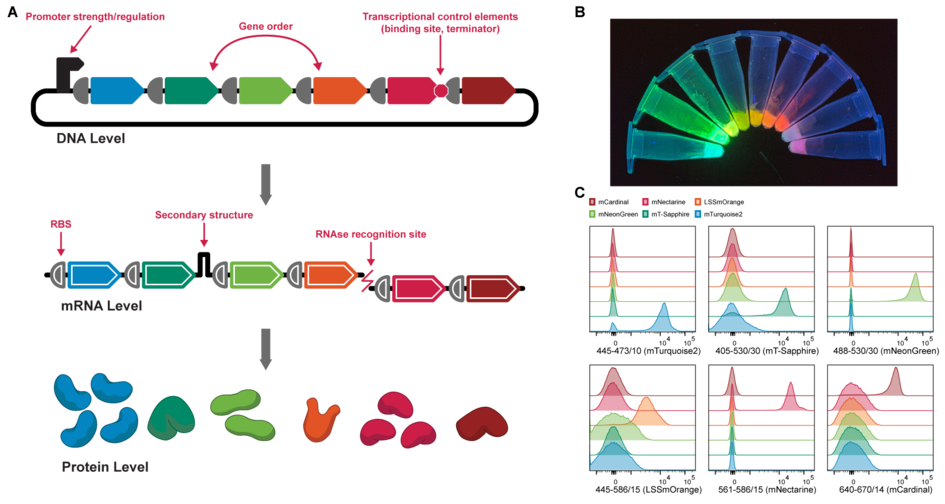
Molecular systems derive their functional breadth from the interplay of multiple elements. The successful cooperation of these elements is often limited to narrow windows of operation, which are often difficult to identify.
We are optimizing complex in vitro systems so they can successfully operate in these windows. For this, we develop cell free systems that allow the synthesis of multiple catalysts and other protein-based elements, and compartmentalize the synthesis in nanoliter or picoliter-sized droplets. This helps us to investigate thousands of system compositions per minute. We use this to develop design rules for multi-membered systems and prepare such droplets for analysis in a classical way (i.e., by fluorescence) and by label-free methods, such as mass-spectrometry. This way we can optimize system function for a variety of objectives, ranging from enzyme evolution to the engineering of smart systems for metabolic diseases.
Panke group @D-BSSE