Artificial Protein Scaffolds for Controlled Assembly of Supramolecular Complexes.
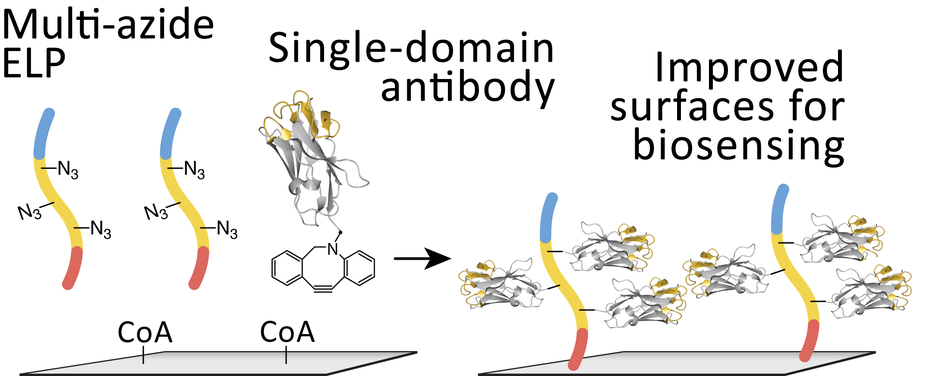
Artificial protein complexes are of high interest for the build-up of molecular factories because they enable assembly of multiple functional subunits in close proximity to one another, providing enhanced catalysis of chemical reactions in series. Assembly of binding proteins (e.g., antibodies) onto scaffolds is also highly desirable for delivery of protein therapeutics because the multi-valent complexes exhibit increased bound lifetime, effectively increasing the apparent affinity of a specific binding molecule to the cell surface.
A major limitation, however, remains the preparation of large scaffolds onto which enzymes and antibodies can assemble. Several strategies have been developed for synthesizing scaffolds, for example, using repeated copies of receptor proteins, or DNA-based scaffolds. These approaches, however, are limited in terms of scaffold size, specificity and/or stability.
The Nash Group synthesizes molecular scaffolds using repetitive protein building blocks called elastin-like polypeptides, and incorporates bioorthogonal functional groups for linkage to proteins and enzymes. The outcomes will be scaffold proteins which provide a versatile platform that is generalizable to many different reaction cascades and binding molecules. This work represents a simplified and scalable process to generate supramolecular complexes with novel functionality.
Nash group @UniBas